Hot Summer, Cool Updates
Summer officially started on June 20th and it’s looking like it’s going to be a scorcher! Rather than focus on the temperature spikes, we’re spending the summer highlighting all things cool – innovations, breakthroughs, entrepreneurs, and more! You’ve heard before that variety is the (sp)ice of life so keep an eye out for features that will feel like a breath of fresh air.
For those that are coming out of quarantine and looking to travel,
average temperatures between June and August are below:
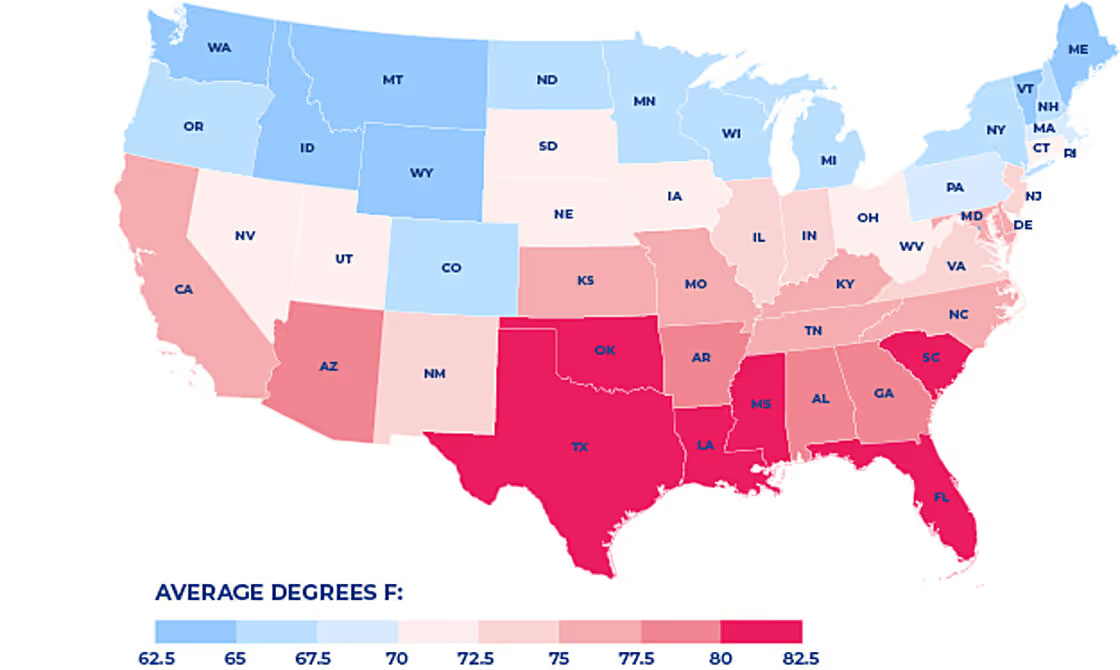
Based on average temperature data for years 2014-2019. Statewide data collected by NOAA National Centers for Environmental information https://www.ncdc.noaa.gov/cag/
Texas topped the charts as the hottest state on average over the past five years – giving us even more motivation to cool things down.
Have ideas of things we should highlight? Share your thoughts! We’d love to hear them.

Help Us, Help You
We started Proxima to demystify the regulatory and clinical research processes and help medical innovators of all sizes and stages. Without a doubt, you have driven us to continue pushing our boundaries to determine new ways for us to help create an accessible, streamlined experience.
Getting a product to market can be confusing and complicated, filled with countless questions and exceptions at each stage. Because of this, we began planning a way for you to quickly get the answers to your burning questions.
We are thrilled to announce the launch of Proxima’s Knowledge Base, which will provide explanations for FAQs and glossary terms. We still have hundreds going through the review process, so check back frequently!
You can now wave goodbye to trudging through countless search engine results to only find confusing answers that require more research just to translate. We’ve got your back!
We also know that we’re still learning about the questions that are most important to you. If there’s something specific you’d like to see added to our Knowledge Base, let us know!

Knowledge Base Highlights:

Have you ever wondered how Medical Device Clinical Trials are categorized? If so, click here to learn more about the 3 types of studies!

Drug sponsors formally propose that the FDA approve a new pharmaceutical through an NDA. But what exactly is an NDA? Find out here!

Words of Wisdom

Medtech Innovator recently interviewed Jaye Thompson, Proxima's co-founder and COO.
Curious about her thoughts, advice, and projections for the future of the industry? Read her whole interview!

Software as a Medical Device?

Does your software as a medical device (SaMD) function properly? Does it meet the needs of the end user? What are the expectations for post-market clinical surveillance and real world evidence for SaMD products in particular? Check out Kevin Coker’s latest podcast. Great tips in there.

Meet the Innovator

We love innovators and especially those with interesting devices. Check out Alec Santiago's interview with Alton Reich, founder and CEO of Vital Metrix, where he details his experience developing an award-winning cardiac health solution.

You're Somebody's Type
Did you know that in the US, someone needs a blood transfusion every two seconds? Unfortunately, due to widespread lockdowns, blood donations have slowed and many areas are experiencing shortages. Many blood centers are also collecting convalescent plasma from patients who have recovered from a case of COVID-19. Antibodies present may help fight the disease and help other patients recover.
For our Houston locals, Gulf Coast Regional Blood Center is accepting donations of both blood and convalescent plasma. They're scheduling appointments based on social distancing protocol to ensure that you can donate safely.
Reading from afar? Find out your nearest donation site.

TMCx Fall 2020 Cohort - Apply Now!
If your Healthcare startup is looking to form enterprise relationships with the country's top healthcare institutions, it might be worth your while to check out the application for TMCx's Fall 2020 cohort.
The application deadline is July 19 - learn more before it passes!

Catch us at our next event! We'd love to see you.





